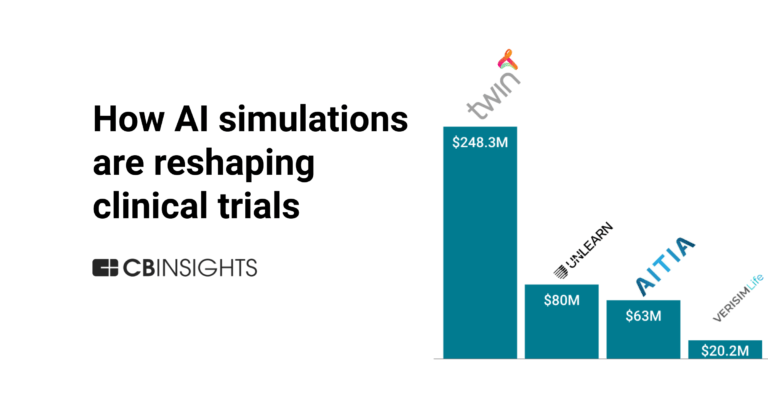
Unlearn
Founded Year
2017Stage
Series C | AliveTotal Raised
$130MValuation
$0000Last Raised
$50M | 2 yrs agoMosaic Score The Mosaic Score is an algorithm that measures the overall financial health and market potential of private companies.
+34 points in the past 30 days
About Unlearn
Unlearn focuses on artificial intelligence and digital twin technology within the clinical research sector. The company provides a platform that creates digital twins of clinical trial participants to predict outcomes and streamline the trial process. Unlearn's solutions aim to improve clinical trials by offering insights and decision support. It was founded in 2017 and is based in San Francisco, California.
Loading...
Unlearn's Product Videos


ESPs containing Unlearn
The ESP matrix leverages data and analyst insight to identify and rank leading companies in a given technology landscape.
The in vivo simulations software market consists of companies developing software that predict drug behavior in living systems. By integrating technology such as AI with pharmacokinetic/pharmacodynamic modeling and biological simulations, these platforms forecast drug movement and interaction within the body. These solutions assist pharmaceutical companies in optimizing drug design, dosing strateg…
Unlearn named as Highflier among 12 other companies, including Certara, Simulations Plus, and CytoReason.
Unlearn's Products & Differentiators
Digital Twins
We build disease-specific AI models trained on extensive, patient-level historical data that generate digital twins of clinical trial participants. Digital twins are comprehensive, longitudinal forecasts of trial participants' clinical outcomes, and enable sponsors to design and run faster trials and earlier go/no-go decisions.
Loading...
Research containing Unlearn
Get data-driven expert analysis from the CB Insights Intelligence Unit.
CB Insights Intelligence Analysts have mentioned Unlearn in 15 CB Insights research briefs, most recently on Sep 25, 2025.

Sep 25, 2025 report
Book of Scouting Reports: AI in Preclinical Development
Aug 22, 2025 report
Book of Scouting Reports: Generative AI in Healthcare & Life Sciences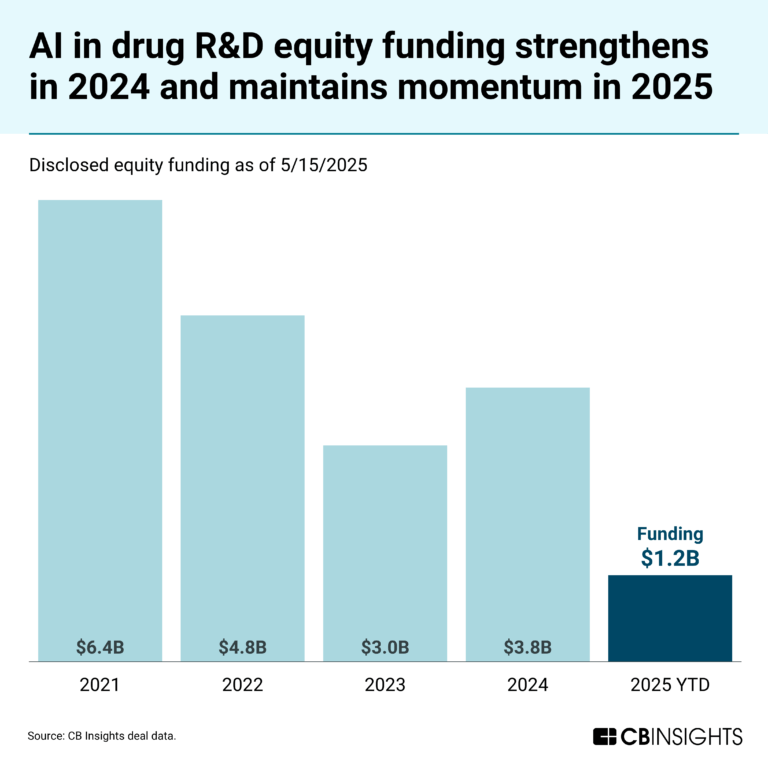
May 23, 2025
The AI in drug R&D market map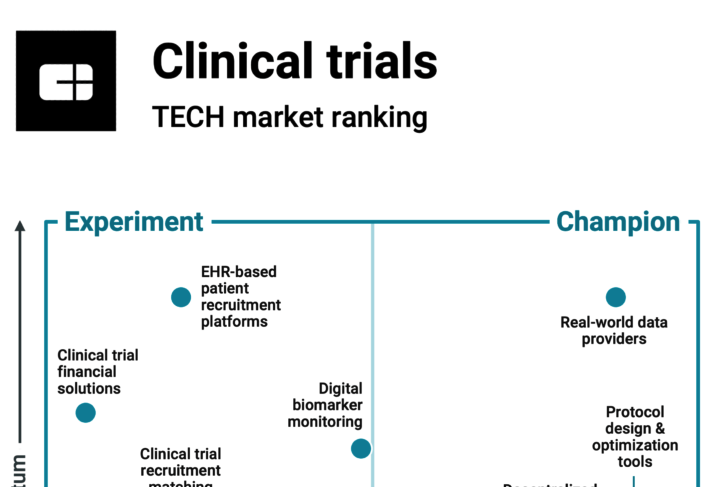

Aug 21, 2024
The clinical trials tech market map
Jan 11, 2024
The generative AI in healthcare market mapExpert Collections containing Unlearn
Expert Collections are analyst-curated lists that highlight the companies you need to know in the most important technology spaces.
Unlearn is included in 7 Expert Collections, including Conference Exhibitors.
Conference Exhibitors
5,302 items
Digital Health 50
300 items
The winners of the second annual CB Insights Digital Health 150.
Digital Health
12,122 items
The digital health collection includes vendors developing software, platforms, sensor & robotic hardware, health data infrastructure, and tech-enabled services in healthcare. The list excludes pureplay pharma/biopharma, sequencing instruments, gene editing, and assistive tech.
AI 100 (All Winners 2018-2025)
100 items
Winners of CB Insights' 5th annual AI 100, a list of the 100 most promising private AI companies in the world.
Smart Cities
1,148 items
Precision Medicine Tech Market Map
160 items
This CB Insights Tech Market Map highlights 160 precision medicine companies that are addressing 9 distinct technology priorities that pharmaceutical companies and healthcare providers face.
Unlearn Patents
Unlearn has filed 21 patents.
The 3 most popular patent topics include:
- machine learning
- artificial neural networks
- design of experiments

Application Date | Grant Date | Title | Related Topics | Status |
|---|---|---|---|---|
8/19/2020 | 7/30/2024 | Artificial neural networks, Machine learning, Design of experiments, Electronic health records, Classification algorithms | Grant |
Application Date | 8/19/2020 |
|---|---|
Grant Date | 7/30/2024 |
Title | |
Related Topics | Artificial neural networks, Machine learning, Design of experiments, Electronic health records, Classification algorithms |
Status | Grant |
Latest Unlearn News
Oct 15, 2025
Expected to grow to $4.18 billion in 2029 at a compound annual growth rate (CAGR) of 28.4%” — The Business Research Company LONDON, GREATER LONDON, UNITED KINGDOM, October 15, 2025 / EINPresswire.com / -- "Get 20% Off All Global Market Reports With Code ONLINE20 – Stay Ahead Of Trade Shifts, Macroeconomic Trends, And Industry Disruptors What Is The Projected Market Size & Growth Rate Of The Virtual Clinical-Trial Participant Avatars Market? The market for virtual participant avatars in clinical trials has seen significant growth in the past few years. This industry is projected to expand from $1.19 billion in 2024 to $1.54 billion in 2025, boasting a compound annual growth rate (CAGR) of 28.8%. Factors leading to such growth during the historic period include the increasing embrace of decentralized clinical trials, growing requirement for diverse patient representation in trials, expansion of wearable health monitoring device usage, regulatory backing for digital trial solutions, and the urgency to shorten clinical trial durations. The market size for avatars used in virtual clinical trials is predicted to witness a massive surge in growth in the upcoming years, reaching a value of $4.18 billion by 2029, with a compound annual growth rate (CAGR) of 28.4%. This significant growth during the forecast period can be credited to the progress in AI-driven patient simulation, the convergence of virtual avatars with electronic data capture systems, the growing emphasis on personalized medicine, increased investments in digital twin technologies, and the broadening of virtual trials for rare diseases and chronic ailments. Key trends anticipated during this period include applying AI-powered digital twins for patient simulation, the incorporation of real-life data and electronic health records, the use of machine learning to predict trial outcomes, the creation of immersive VR/AR platforms for enhancing patient engagement, and the employment of blockchain for the secure management of patient data. Download a free sample of the virtual clinical-trial participant avatars market report: https://www.thebusinessresearchcompany.com/sample.aspx?id=28351&type=smp What Is The Crucial Factor Driving The Global Virtual Clinical-Trial Participant Avatars Market? The expansion of the virtual clinical-trial participant avatars market is anticipated to be driven by the increasing number of clinical trials. Clinical trials, which are structured studies designed to assess the safety and effectiveness of medical interventions in human subjects, are on the rise due to amplified pharmaceutical investment in pharmaceutical development. This is due to the quest for novel treatments for complicated illnesses and personalized medicine methods. Avatars of clinical-trial participants generate AI-enabled digital patient profiles that mimic disease progression, treatment reaction, and safety results. These assist in quicker recruitment, intelligent trial design, earlier detection of effective therapies, and minimized expenses and risks. For instance, as reported by the Association of the British Pharmaceutical Industry, a trade organization based in the UK in November 2023, the total number of industry-funded clinical trials initiated in the UK annually rose modestly by 4.3%, escalating from 394 trials in 2021 to 411 trials in 2022. Hence, the escalation in the number of clinical trials is spurring the growth of the virtual clinical-trial participant avatars market. Who Are The Emerging Players In The Virtual Clinical-Trial Participant Avatars Market? Major players in the Virtual Clinical-Trial Participant Avatars Global Market Report 2025 include: • NVIDIA Corporation • Dassault Systèmes SE • Signant Health Holding Corp. • Owkin Inc • Medable Inc. • Insilico Medicine Inc. • Science 37 Inc. • THREAD Inc. • Lindus Health Limited • Obvio Health What Are The Main Trends, Positively Impacting The Growth Of Virtual Clinical-Trial Participant Avatars Market? Major corporations in the virtual clinical trial participant avatars sector are turning their attention towards the development of cutting-edge solutions, such as digital twin-based clinical trial participants. These innovative solutions aim to boost the effectiveness of trials and enhance the precision in predicting drug development outcomes. By simulating real patients, digital twin-based clinical trial participants replicate their physiological and behavioral reactions, allowing researchers to predict the results of treatments and fine-tune trial designs, thereby decreasing the need for extensive physical testing. An illustration of this innovation is the update launched by the US-based AI clinical trial technology firm, Unlearn.ai, Inc., in July 2023. They unveiled an updated digital twin generator (DTG) for Alzheimer's disease, 3.1 version, which significantly improves the handling of missing baseline data by incorporating a neural network imputation model within a Neural Boltzmann Machine structure. This led to less sensitivity towards missing data and an overall superior performance. The DTG draws upon various patient data sets to generate individualized digital twins that can accurately forecast clinical and biomarker trajectories, thereby aiding clinical trials with a more exact and robust modeling of Alzheimer's progression. What Segments Are Covered In The Virtual Clinical-Trial Participant Avatars Market Report? The virtual clinical-trial participant avatars market covered in this report is segmented as 1) By Avatar Type: Artificial Intelligence-Generated Avatars, Digitally Modeled Human Avatars, Hybrid Avatars 2) By Deployment Mode: Cloud-Based, On-Premises 3) By Application: Drug Development, Patient Recruitment, Data Simulation, Remote Monitoring, Other Applications 4) By End-User: Pharmaceutical Companies, Contract Research Organizations, Academic And Research Institutes, Other End-Users Subsegments: 1) By Artificial Intelligence-Generated Avatars: Machine Learning-Based Avatars, Deep Learning-Based Avatars, Natural Language Processing (NLP)-Enabled Avatars, Generative Adversarial Network (GAN)-Based Avatars 2) By Digitally Modeled Human Avatars: Three-Dimensional (3D) Scanned Human Avatars, Motion Capture Modeled Avatars, Photorealistic Rendered Avatars, Anatomically Accurate Human Avatars 3) By Hybrid Avatars: artificial intelligence (AI)-Enhanced Digitally Modeled Avatars, Mixed Reality (MR) Enabled Avatars, Cognitive Simulation Avatars, Sensor-Integrated Virtual Avatars View the full virtual clinical-trial participant avatars market report: https://www.thebusinessresearchcompany.com/report/virtual-clinical-trial-participant-avatars-global-market-report Which Region Is Projected To Hold The Largest Market Share In The Global Virtual Clinical-Trial Participant Avatars Market? In the year 2024, the Virtual Clinical-Trial Participant Avatars Global Market Report indicated North America as the dominant region. The projected growth outlook suggests that Asia-Pacific will experience the quickest expansion during the forecast period. The report incorporates a thorough examination of regions including Asia-Pacific, Western Europe, Eastern Europe, North America, South America, the Middle East, and Africa. Browse Through More Reports Similar to the Global Virtual Clinical-Trial Participant Avatars Market 2025, By The Business Research Company Virtual Clinical Trials Global Market Report 2025 https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report Patient Simulators Global Market Report 2025 https://www.thebusinessresearchcompany.com/report/patient-simulators-global-market-report Virtual Human Anatomy Software Global Market Report 2025 https://www.thebusinessresearchcompany.com/report/virtual-human-anatomy-software-global-market-report Speak With Our Expert: Saumya Sahay Americas +1 310-496-7795 Asia +44 7882 955267 & +91 8897263534 Europe +44 7882 955267 Email: saumyas@tbrc.info The Business Research Company - www.thebusinessresearchcompany.com Follow Us On: • LinkedIn: https://in.linkedin.com/company/the-business-research-company Oliver Guirdham The Business Research Company info@tbrc.info Visit us on social media: LinkedIn Facebook X Legal Disclaimer: EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Unlearn Frequently Asked Questions (FAQ)
When was Unlearn founded?
Unlearn was founded in 2017.
Where is Unlearn's headquarters?
Unlearn's headquarters is located at 303 2nd Street, San Francisco.
What is Unlearn's latest funding round?
Unlearn's latest funding round is Series C.
How much did Unlearn raise?
Unlearn raised a total of $130M.
Who are the investors of Unlearn?
Investors of Unlearn include DCVC, 8VC, Radical Ventures, Wittington Ventures, EPIC Ventures and 12 more.
Who are Unlearn's competitors?
Competitors of Unlearn include InSilicoTrials, Ingenix, Perceiv AI, Aitia, January and 7 more.
What products does Unlearn offer?
Unlearn's products include Digital Twins.
Loading...
Compare Unlearn to Competitors

Nova In Silico specializes in the development of a clinical trial simulation platform within the biotechnology and pharmaceutical industry. Their main offering, jinkō, is a tool that streamlines clinical trials by using disease models and digital patients to predict clinical outcomes and optimize trial design. The platform is primarily utilized by biotech and pharma companies, academic research centers, and not-for-profit organizations to accelerate and de-risk the drug development process. It was founded in 2010 and is based in Lyon, France.

My Intelligent Machines (MIM) specializes in artificial intelligence and systems biology within the biotechnology sector. The company offers augmented intelligence systems and an intelligent platform designed to assist in early drug development, enabling precise biological simulations and the development of targeted and personalized therapies. Its systems are primarily marketed to the biopharma industry, supporting patient stratification, therapeutic target selection, and biomarker identification for more efficient and personalized therapies. It was founded in 2016 and is based in Montreal, Canada.

PhaseV provides artificial intelligence (AI) and machine learning solutions for clinical development within the biopharmaceutical sector. The company offers tools for optimizing clinical trials by utilizing advanced analytics and predictive modeling. PhaseV primarily serves biopharma sponsors and contract research organizations (CROs) looking to address challenges in trial success rates and time-to-market. PhaseV was formerly known as Phavev Trials Ltd.. It was founded in 2023 and is based in Cambridge, Massachusetts.

Cytocast is a TechBio startup focused on advancing drug development through innovative simulation and modeling technologies. The company offers the Cytocast Simulated Cell, a platform designed to predict drug side effects by simulating molecular interactions within human cells, and the Cytocast Digital Twin technology, which aims to personalize treatments based on individual molecular characteristics. It was founded in 2019 and is based in Budapest, Hungary.

Mostly AI specializes in creating synthetic data within the technology and data security sectors. The company offers a platform and an open-source Software Development Kit (SDK) that enables organizations to generate, analyze, and share synthetic data, allowing for data insights and collaboration. Mostly AI's solutions support applications such as machine learning (AI) machine learning (ML) model training, software testing, and data sharing, while addressing privacy regulations. It was founded in 2017 and is based in Vienna, Austria.

AICURA medical focuses on the intersection of data and artificial intelligence within the healthcare and life sciences sectors. The company provides a platform for the integration, curation, and analysis of multimodal clinical data, using pre-trained AI models for clinical research and patient outcomes. AICURA medical serves biotech firms, healthcare providers, medtech companies, clinical researchers, and contract research organizations (CROs), contributing to drug development and patient care. It was founded in 2017 and is based in Berlin, Germany.
Loading...